Biomed Briefs
Strong Heart Study in 7th phase
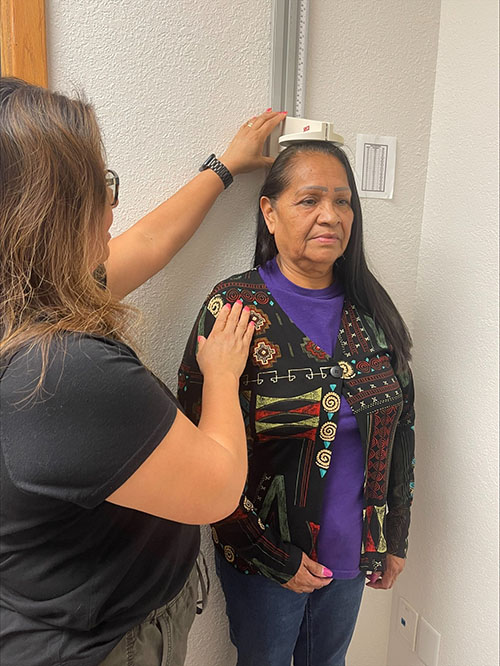

Launched in the 1980s, the Strong Heart Study is the longest-running study of cardiovascular health in American Indians. About every five years, local research centers work with tribes in Arizona, Oklahoma and the Dakotas to collect data from participants. The seventh phase is seeking to screen about 2,600 participants over two years, including all living members of the original cohort. “It is a very big undertaking, both for the field staff and for the participants,” says Texas Biomed Professor Shelley Cole, PhD, chair of the study steering committee. “We appreciate how participants are taking the time to provide their samples and trusting the researchers with them.” Along with a basic health exam, including height, weight, blood pressure, blood and urine samples, additional screenings aim to learn more about topics like mental health or microbiomes. Long-term studies like this provide valuable insights into how health is influenced by a wide range of factors, from genetics to environment.
CoDe tool makes vaccine development faster and more accurate

A new software tool developed by Texas Biomed Professor Luis Martinez-Sobrido, PhD, and collaborators in India and Spain can help scientists and vaccine developers quickly edit genetic blueprints of pathogens to make them less harmful. The tool, called CoDe – short for Codon Deoptimization – enables users to make precise edits to a genetic code to make genes less functional. Often, vaccine developers want to optimize or turn up expression of certain genes, and there are plenty of software tools to help with that. But for live attenuated vaccines that contain the full genetic sequence of a virus, it is critical to deoptimize, or turn down, certain genes to make the vaccines safe and effective. “With CoDe we can precisely mutate thousands of nucleotides in a single gene,” Dr. Martinez-Sobrido says. “It also enables us to modulate gene expression of small sections, rather than knocking out entire genes completely. That is very helpful for vaccine design as well as basic research.” CoDe is freely available for other academic users and available to license by industry partners.
Therapy shows promise to help clear tuberculosis
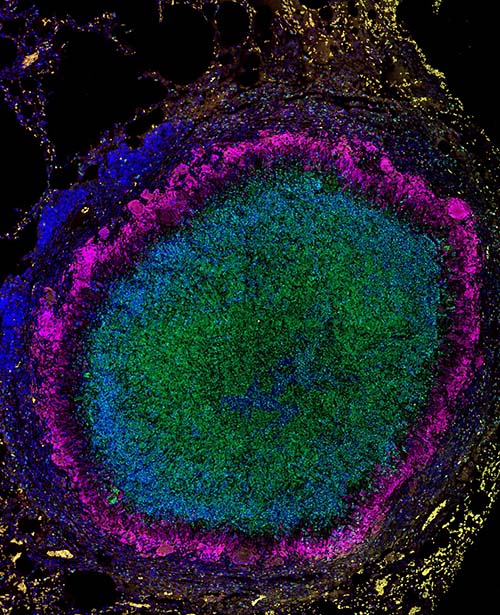

A team of Texas Biomed and SNPRC scientists, led by Professor Smriti Mehra, PhD, have identified a promising way to help fight tuberculosis (TB), a disease that still kills nearly 2 million people annually. The research focuses on a potential host-directed therapy that can bolster the body’s ability to control infection. Specifically, the team found inhibiting an enzyme known as IDO – short for Indoleamine 2,3 dioxygenase – helped nonhuman primates better eliminate active TB infection. Blocking IDO for four weeks in conjunction with antibiotics led to improved health metrics compared with antibiotics alone. “This is exciting,” says Dr. Mehra. “We have promising results suggesting an IDO inhibitor could be a host-directed therapy that reduces the length of time and amount of antibiotics that TB patients have to take, especially those with multidrug resistance.”
Solving a piece of the TB puzzle
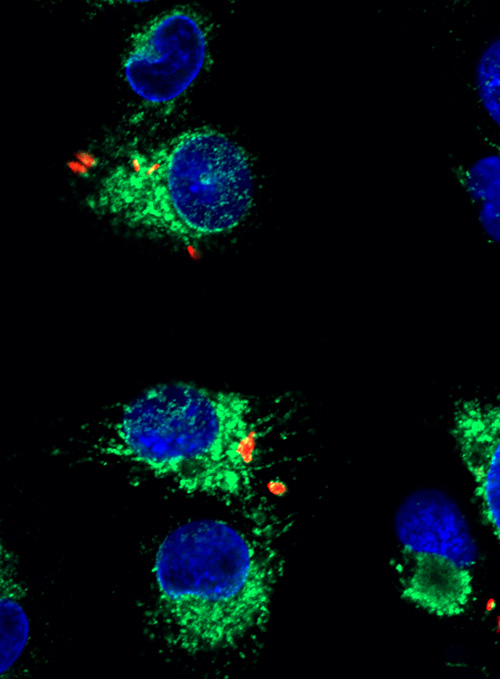

Like a biological trojan horse, Mycobacterium tuberculosis (M.tb), the pathogen responsible for tuberculosis, infects and hides inside macrophages, the immune system’s first responders. M.tb hijacks macrophages’ cell signaling pathways, evading detection and promoting its own growth. Texas Biomed Staff Scientist Chrissy Leopold Wager, PhD, has elucidated some of the mechanisms behind the bacteria’s prowess. Working with human macrophages, Dr. Leopold Wager and colleagues in Professor Larry Schlesinger’s lab identified the precise pathway M.tb uses to activate CREB, a critical protein involved with DNA transcription. CREB is like a conductor, deciding the time and level of expression of very specific genes, setting in motion precise cascades of reactions. They found that activation of CREB dampens the inflammatory response inside macrophages; specifically, it prevents the fusion of vesicles containing M.tb with lysosomes, little pockets of acids that would otherwise help destroy the bacteria. Excitingly, Dr. Leopold Wager found that inhibiting CREB allows fusion to proceed and helps control M.tb growth. The study, published in PLOS Pathogens, identifies CREB as a new and interesting target to combat tuberculosis.
Prior dengue infection may make Zika outcomes worse


Researchers at Texas Biomed and the Trudeau Institute in New York have discovered that female marmosets are more likely to transmit the Zika virus during pregnancy if they have been previously infected by a different virus, dengue. The research, published in Science Translational Medicine, may help unravel why the 2015 Zika outbreak led to thousands of miscarriages, stillbirths and cases of permanent birth defects. Texas Biomed Professor Corinna Ross, PhD, Professor Emeritus Jean Patterson, PhD, and their collaborators found that pregnant marmoset monkeys that had dengue previously passed 100,000 times the amount of Zika virus to their fetuses than marmosets that had never had dengue. The researchers suspect that Zika is using dengue antibodies to cross the placenta. Additional research is needed to fully understand the mechanisms and if the findings hold true for humans.
Second Gene Implicated In Malaria Drug Resistance

The story of malaria’s resistance to the antimalarial drug chloroquine has long been thought to involve only one key gene. An international team led by Texas Biomed, University of Notre Dame, Seattle Children’s Hospital and the Medical Research Council Unit The Gambia, found a second gene is also involved. Researchers analyzed 600 genomes of Plasmodium falciparum, the deadliest species of malaria parasite, collected from 1984 to 2004. The dataset revealed mutations in another gene increased from 0% to 97% frequency over 30 years – clear signs of natural selection in action. A variety of additional analyses and experiments confirmed the mutations help confer drug resistance. “With drug-resistant pathogens on the rise, it is important to understand how treatments drive parasite evolution and how this evolution can vary in different parts of the world,” says Texas Biomed Professor Timothy J.C. Anderson, PhD. The research was published in Nature Microbiology.
“No More Schisto” Game Wins International Award


Texas Biomed postdoctoral researcher Egie Enabulele, PhD, won an international award for a game he developed about the devastating parasitic disease, schistosomiasis, and how to avoid becoming infected. “No More Schisto” was recognized by The International Society of Neglected Tropical Diseases (ISNTD) with the ISNTD Festival 2023 Game Award. The board game can be printed on an A4 letter size piece of paper and is aimed at elementary school-aged children, who can become infected by swimming in lakes and rivers where the parasites and their snail vectors are present. “I wanted to design something that can be distributed and played in the field, and that could be easily translated into other languages,” says Dr. Enabulele, whose own kids and friends helped him test out and improve the game. Passionate about art and making science understandable for kids, he plans to make games about other diseases as well. “Education and behavior changes are a crucial strategy for eliminating many neglected tropical diseases,” he says.
HIV vaccine candidate aims to block virus before it takes root
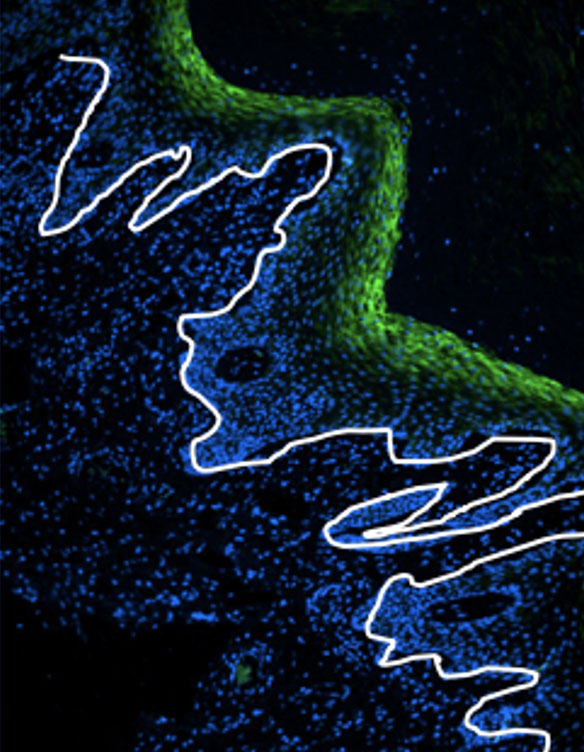

The National Institutes of Health awarded $3.8 million to Texas Biomed Professor Marie-Claire Gauduin, PhD, to further develop a promising HIV vaccine candidate over the next four years. An effective HIV vaccine has eluded the biomedical community for decades, in part because the virus spreads throughout the body within weeks of infection. Dr. Gauduin is aiming to stop HIV where it enters by designing a vaccine that specifically targets the interior lining of the vagina and rectum, called the mucosal epithelium. The vaccine stimulates the production of antibodies in these areas. Much like a bouncer checking for fake IDs just inside the door, or sentries along a castle wall, the antibodies are well positioned to quickly respond and block the virus from proceeding any further. The grant will allow Dr. Gauduin and her team to build on promising results in nonhuman primates and gather more data about mechanisms, safety, efficacy and different delivery methods. “I am excited to move forward and hope to get to the preclinical stage for human trials,” Dr. Gauduin says.